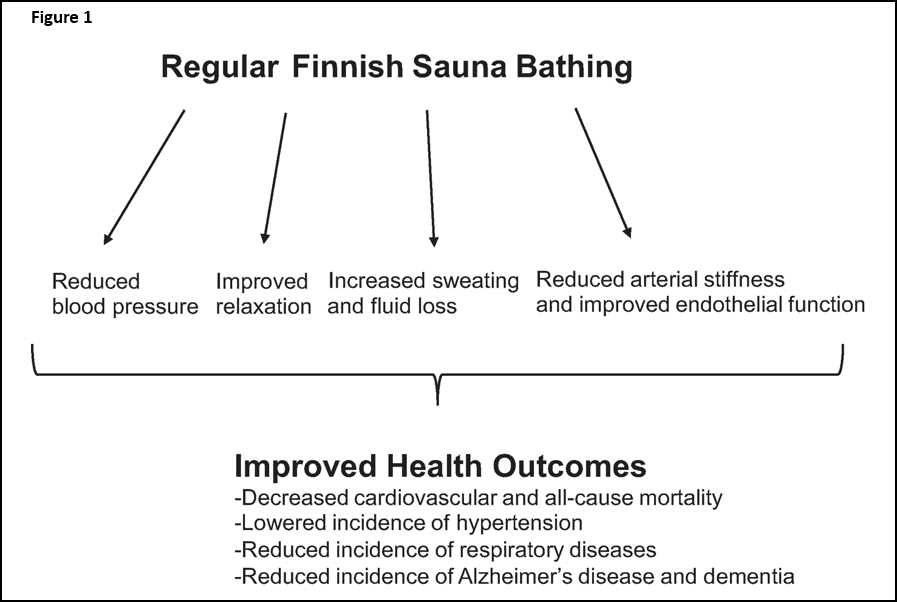
Using Saunas for Detoxification
We recently talked about how important glutathione is for detoxification, and in this blog, we are going to talk about another important tool in our detox toolbox: The sauna. A sauna induces a state in the body called hyperthermia (raised body temperature), which increases peripheral blood circulation through the induction of thermal stress in the body. When heat challenges the body, the body compensates by directing blood to the skin, inducing sweating, reduced blood pressure, and increased heart rate1. There are several types of saunas: Scandinavian saunas (dry heat, 40-60% humidity), Roman baths, or infrared (IR) saunas. This last sauna directs IR rays to heat exposed skin tissue2. Some of the documented benefits include reduced blood pressure, reduced arterial stiffness, decreased risk of cardiovascular disease, improved relaxation, and others3-8.(See Figure 1)
I won’t compare these different types of saunas because their use and efficacy depend on the situation. Factors that go into this decision are the condition of the patient, what chemicals are you trying to detox, and what is available. In this article, I’m going to talk about how some studies demonstrated the utility of saunas in treatment.
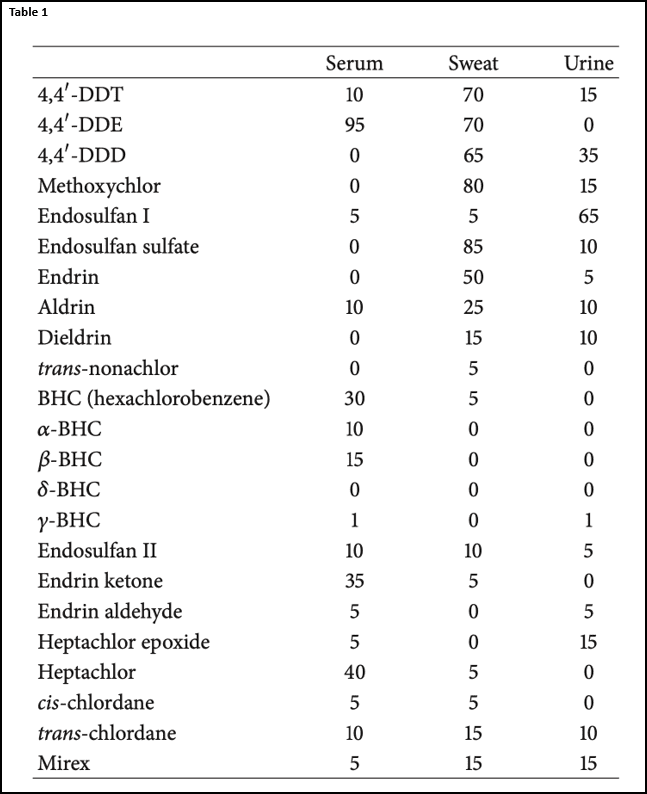
The first type of compounds I’m going to discuss are organochlorinated pesticides (OCPs). OCPs accumulate in body tissues and have long half-lives (up to 15 years)9. OCPs are lipophilic, which means that they are easily absorbed in fat and other tissues such as the skin. OCPs have a low vapor pressure and degrades slowly in the environment10. These OCPs cause mitochondrial damage, oxidative stress, endocrine disruption, and dysregulation of synapses by altering the cation channels11-13. One of the best papers discussing the excretion of OCPs was from Stephen Genuis in 2016. This paper demonstrated that many OCPs come out better in sweat than urine, except for endosulfan14. (Table 1) From these results it seems DDT, DDE, DDD, methoxychlor, endosulfan, and endrin can be induced out of the body through perspiration. Because these toxins are lipophilic, you do not see them readily detected in blood (similar to mycotoxins). However, it is encouraging that these toxins can be perspired, which means that perhaps mycotoxins could be perspired as well.
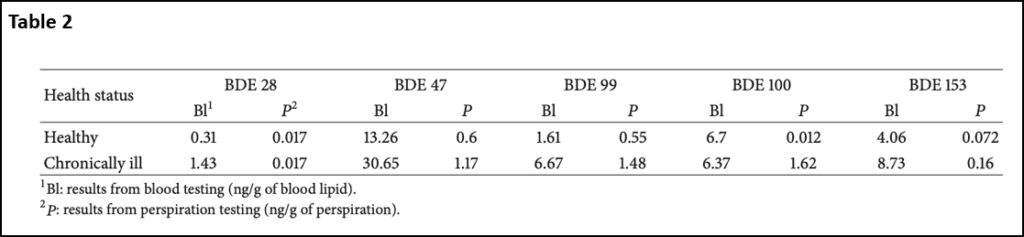
The next set of compounds that I wanted to cover are polybrominated diphenyl ethers (PBDEs). There are 209 different types of PBDEs that are used in applications such as foam, textiles, plastics, electrical equipment, computers, and construction materials15. Since these are not fixed into polymer matrices, these PBDEs can leak out and spread into the environment. They have proven to be bioaccumulating and detectable in air, soil, wildlife, and humans16-19. The PBDEs are classified by numbers 1 through 209 and the most common versions seen in the environment are 28, 47, 99, 100, and 15320. A study looking at blood and perspiration of these chemicals found that chronically ill patients had higher levels than a healthy control group (Table 2). BDE28 and BDE99 had levels five times higher in chronically ill patients. BDE47 had values three times higher in chronically ill patients. BDE47 had the highest value in the blood at 30.54 ng/g of blood lipid. For many of these toxins, there was a correlation between the blood amount and the amount seen in perspiration, indicating that sauna may be a valuable tool for detoxifying these chemicals20.
Although there are still limited studies on the use of saunas for the purpose of detox, the limited results out there are encouraging for multiple types of toxins. Obviously, the easiest types of toxins to eliminate in this manner are those more water soluble, however, data from these last two studies shows that some lipophilic toxins can also be eliminated through sweat. The RealTime Environmental Pollutant Profile can help you to see if you are eliminating a larger than normal amount of multiple toxins. This can also be run along with our Glyphosate Exposure Analysis.
1. C. G. Crandall, J. Gonzalez-Alonso, Cardiovascular function in the heat-stressed human. Acta Physiol (Oxf) 199, 407-423 (2010).
2. K. H. Cho et al., Effect of water filtration infrared-A (wIRA) sauna on inorganic ions excreted through sweat from the human body. Environ Sci Pollut Res Int 30, 18260-18267 (2023).
3. T. Laukkanen, J. A. Laukkanen, S. K. Kunutsor, Sauna Bathing and Risk of Psychotic Disorders: A Prospective Cohort Study. Med Princ Pract 27, 562-569 (2018).
4. J. A. Laukkanen, T. Laukkanen, S. K. Kunutsor, Cardiovascular and Other Health Benefits of Sauna Bathing: A Review of the Evidence. Mayo Clin Proc 93, 1111-1121 (2018).
5. S. K. Kunutsor, T. Laukkanen, J. A. Laukkanen, Longitudinal associations of sauna bathing with inflammation and oxidative stress: the KIHD prospective cohort study. Ann Med 50, 437-442 (2018).
6. S. K. Kunutsor et al., Short-term effects of Finnish sauna bathing on blood-based markers of cardiovascular function in non-naive sauna users. Heart Vessels 33, 1515-1524 (2018).
7. M. Kallstrom et al., Effects of sauna bath on heart failure: A systematic review and meta-analysis. Clin Cardiol 41, 1491-1501 (2018).
8. T. Laukkanen, H. Khan, F. Zaccardi, J. A. Laukkanen, Association between sauna bathing and fatal cardiovascular and all-cause mortality events. JAMA Intern Med 175, 542-548 (2015).
9. U. N. E. Programme (Stockholm Convention: Proteching health and the environment from persistent organic pollutants
10. W. P. James, R. S., Principles of Toxicology (John Wiley & Sons, New York, NY, USA, 2000).
11. B. Hille, Pharmacological modifications of the sodium channels of frog nerve. J Gen Physiol 51, 199-219 (1968).
12. W. Mnif et al., Effect of endocrine disruptor pesticides: a review. Int J Environ Res Public Health 8, 2265-2303 (2011).
13. R. Pathak et al., Organochlorine pesticide residue levels and oxidative stress in preterm delivery cases. Hum Exp Toxicol 29, 351-358 (2010).
14. S. J. Genuis, K. Lane, D. Birkholz, Human Elimination of Organochlorine Pesticides: Blood, Urine, and Sweat Study. Biomed Res Int 2016, 1624643 (2016).
15. S. C. Secretariat (2008) “The POPs” Stockholm Convention. .
16. P. O. Darnerud, G. S. Eriksen, T. Johannesson, P. B. Larsen, M. Viluksela, Polybrominated diphenyl ethers: occurrence, dietary exposure, and toxicology. Environ Health Perspect 109 Suppl 1, 49-68 (2001).
17. R. A. Hites, Polybrominated diphenyl ethers in the environment and in people: a meta-analysis of concentrations. Environ Sci Technol 38, 945-956 (2004).
18. R. C. Hale, M. J. La Guardia, E. Harvey, T. M. Mainor, Potential role of fire retardant-treated polyurethane foam as a source of brominated diphenyl ethers to the US environment. Chemosphere 46, 729-735 (2002).
19. J. L. Lyche, C. Rosseland, G. Berge, A. Polder, Human health risk associated with brominated flame-retardants (BFRs). Environ Int 74, 170-180 (2015).
20. S. K. Genuis, D. Birkholz, S. J. Genuis, Human Excretion of Polybrominated Diphenyl Ether Flame Retardants: Blood, Urine, and Sweat Study. Biomed Res Int 2017, 3676089 (2017).
21. I. Heinonen, J. A. Laukkanen, Effects of heat and cold on health, with special reference to Finnish sauna bathing. Am J Physiol Regul Integr Comp Physiol 314, R629-R638 (2018)
