Symptoms of Mold Illness

Mold symptoms are often misdiagnosed or missed entirely, but they can cause real harm. Mycotoxin Toxicity or mycotoxicosis, also commonly referred to as mold illness or mold poisoning, is caused by exposure to mycotoxins, the toxic substances produced by fungi or mold. Mold is all around us and people have largely lived alongside mold in a symbiotic relationship. Some molds, however, are poisonous to humans and our pets. These molds can be found in our homes and in a variety of foods we consume such as our morning coffee, afternoon trail mix, or even cheese we eat.
The severity of the mycotoxin experience depends on 3 main factors:
- The type of mycotoxin you are exposed to
- The extent of your exposure
- Your body’s ability to detoxify
Ultimately, the longer or more intense your exposure is to mycotoxins, the greater the chance you may develop noticeable or even severe symptoms. Additionally, each body handles toxins differently. Genetics, overall health, and even age can influence how effectively someone is able to eliminate mycotoxins. Because of these variations, people exposed to the same volume of mycotoxins may experience entirely different symptoms at varying degrees of intensity.
There are, however, common symptoms that are often overlooked. Dr. Marjorie Peak, a RealTime Labs consulting physician, notes that, “The majority of patients with mold or environmental illness have seen 10-12 doctors before finding the causes and beginning of solutions to their ailments. This is partly because symptoms are nonspecific and overlap other conditions.” The most common mycotoxin toxicity symptoms include:
- Chronic fatigue/exhaustion
- Sinusitis and sinus issues
- Brain fog
- Immune suppression
- Chronic illness
- Depression
- Anxiety
- Anemia
- Fluid retention
- General weakness
- Nausea
- Intestinal distress (irritable bowel, diarrhea, cramps, etc.)
- Asthma
- Hearing problems
- Rashes and hives
- Shortness of breath
- Migraine and headaches
Diagram of Mold Symptoms in the Body
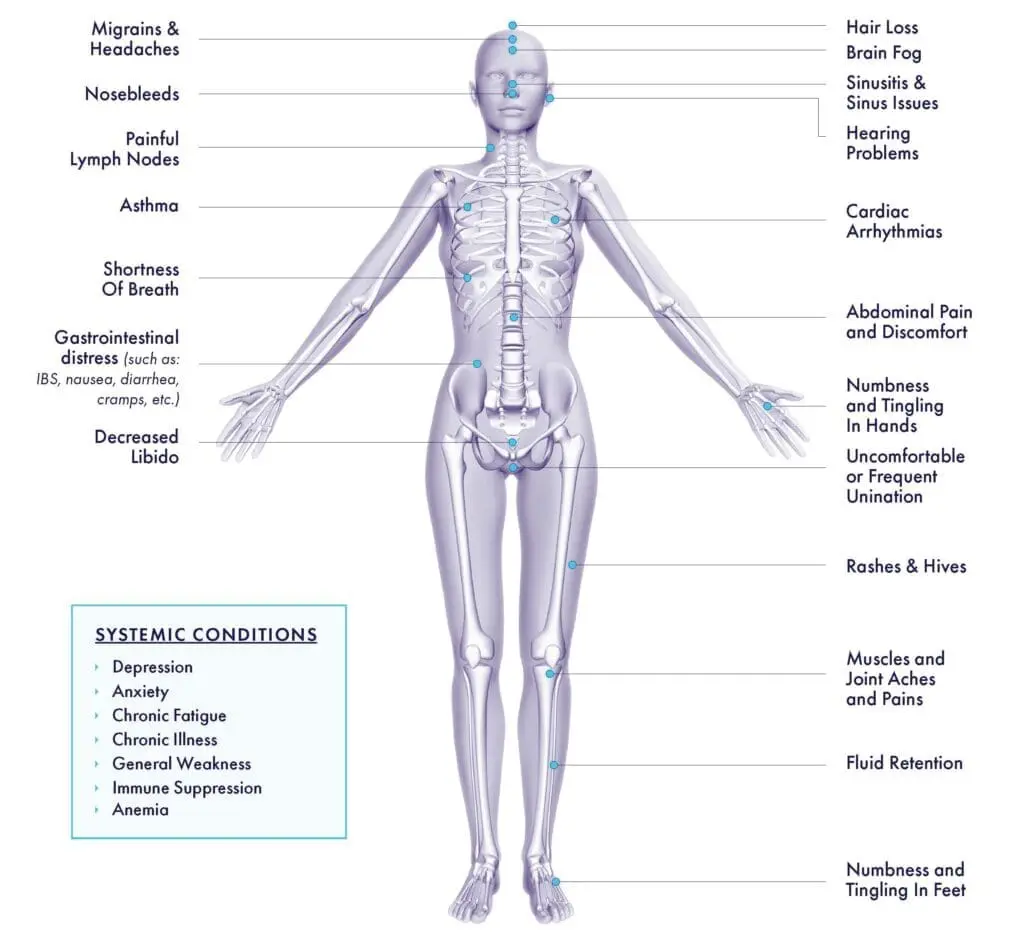
Mold Toxicity symptoms can be nonspecific or appear similar to symptoms caused by inflammatory issues such as food sensitivities, mast cell activation syndrome (MCAS), or even viruses. Therefore, understanding where mycotoxin exposures happen and what your potential exposure may be is imperative. Asking yourself questions like: “Did my symptoms start after a move to a new home or work environment?” can help you identify if mold may be the root cause of symptoms.
For a full list of questions to ask if you think you may be suffering from mold poisoning or mycotoxin toxicity, take a look at our blog “6 Questions To Ask Yourself If You Think You Have Toxic Mold Poisoning”.
The best way to determine if your symptoms or illness is caused by or being inflamed by mycotoxin toxicity is to get tested. A simple urine test can tell you if you have been exposed to 16 of the most common mycotoxins that are toxic to humans. This will give you the insights you need to know if mold is the culprit behind your health issues.
References
- D. W. Holt, Monitoring mycophenolic acid. Ann Clin Biochem 39, 173-183 (2002).
- S. Konig et al., Gliotoxin from Aspergillus fumigatus Abrogates Leukotriene B4 Formation through Inhibition of Leukotriene A4 Hydrolase. Cell Chem Biol 26, 524-534 e525 (2019).
- Y. Jiang et al., Aflatoxin-related immune dysfunction in health and in human immunodeficiency virus disease. Clin Dev Immunol 2008, 790309 (2008).
- Q. Wu et al., Immune Evasion, a Potential Mechanism of Trichothecenes: New Insights into Negative Immune Regulations. Int J Mol Sci 19 (2018).
- W. P. Liew, S. Mohd-Redzwan, Mycotoxin: Its Impact on Gut Health and Microbiota. Front Cell Infect Microbiol 8, 60 (2018).
- J. H. Rosenblum Lichtenstein et al., Environmental mold growth and mycotoxin exposures elicit specific cytokine and chemokine responses. PLoS One 10, e0126926 (2015).
- M. Flamant-Hulin, I. Annesi-Maesano, D. Caillaud, Relationships between molds and asthma suggesting non-allergic mechanisms. A rural-urban comparison. Pediatr Allergy Immunol 24, 345-351 (2013).
- J. Blatter et al., Fungal exposure, atopy, and asthma exacerbations in Puerto Rican children. Ann Am Thorac Soc 11, 925-932 (2014).
- A. A. Inamdar et al., Fungal-derived semiochemical 1-octen-3-ol disrupts dopamine packaging and causes neurodegeneration. Proc Natl Acad Sci U S A 110, 19561-19566 (2013).
- L. D. Empting, Neurologic and neuropsychiatric syndrome features of mold poisoning and mycotoxin exposure. Toxicol Ind Health 25, 577-581 (2009).
- C. B. Purzycki, D. H. Shain, Fungal toxins and multiple sclerosis: a compelling connection. Brain Res Bull 82, 4-6 (2010).
- A. M. Ratnaseelan, I. Tsilioni, T. C. Theoharides, Effects of Mycotoxins on Neuropsychiatric Symptoms and Immune Processes. Clin Ther 40, 903-917 (2018).
- S. Sehata et al., Morphological and microarray analysis of T-2 toxin-induced rat fetal brain lesion. Food Chem Toxicol 42, 1727-1736 (2004).
- M. Chaudhary, P. V. Rao, Brain oxidative stress after dermal and subcutaneous exposure of T-2 toxin in mice. Food Chem Toxicol 48, 3436-3442 (2010).
- V. Sava, O. Reunova, A. Velasquez, R. Harbison, J. Sanchez-Ramos, Acute neurotoxic effects of the fungal metabolite ochratoxin-A. Neurotoxicology 27, 82-92 (2006).
- G. Dirheimer, E. E. Creppy, Mechanism of action of ochratoxin A. IARC Sci Publ, 171-186 (1991).
- D. E. Bredesen et al., Reversal of cognitive decline in Alzheimer’s disease. Aging (Albany NY) 8, 1250-1258 (2016).
