Home testing for mold and mycotoxin
Welcome back to the RTL blog. Recently, I discussed what symptoms could indicate exposure to significant levels of mycotoxins. For patients with toxic levels of mycotoxins in their bodies, it’s equally important to minimize new exposure as it is to pull out the current toxins in the body. Through my studies and those of other scientists, it seems the most common exposure is environmental from either a home, workplace, or school. The second most significant is what has been labeled either a mold colonization or infection (I will discuss this more in a later blog post), and the least significant of the three is food (I will also discuss this in a later blog). In this blog, I will detail what to consider about home testing.
What can cause the growth of mycotoxins in the home?
There are multiple factors that will influence the growth of mycotoxins. Some of these include the temperature, humidity, the type of surface, and most importantly, how much moisture is available. The humidity and type of surface are factors that we have the most control over. Here is a table from a study looking at mold growth in a water-damaged building.
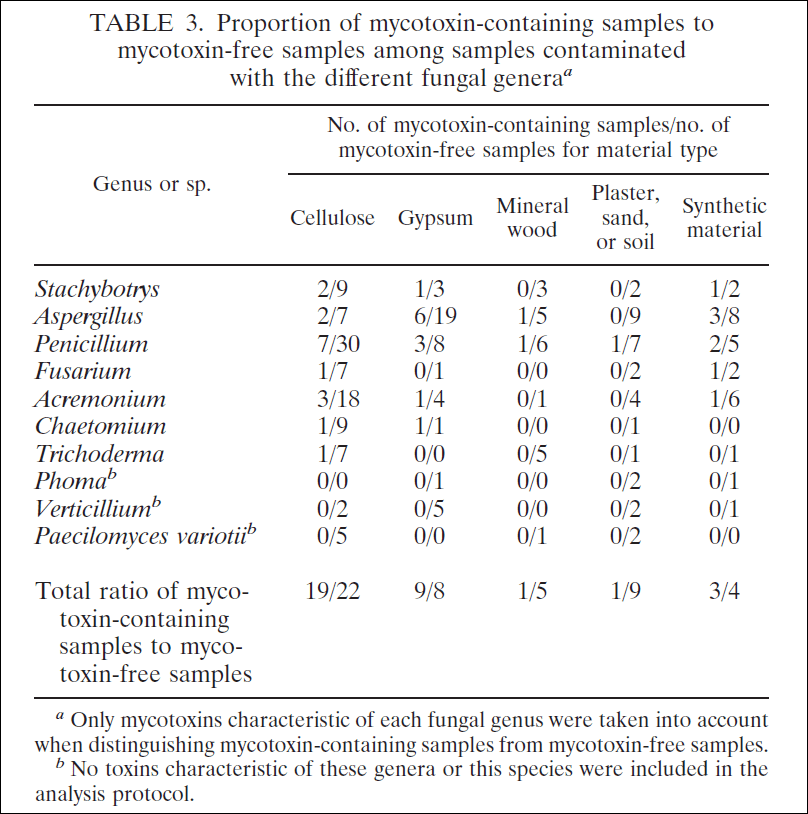
The takeaway from this study is that homes with walls that are made up of cellulose, synthetic material, or gypsum have a lot more mold and mycotoxins than homes made of plaster or mineral wood. The conclusion from this study seems counterintuitive, that older homes are safer than newer homes. And that is exactly the feedback I’m getting from the remediation community, that they are seeing more moldy homes in newer builds than older homes. Newer homes are more often built with synthetic material and drywall, where older homes more often have plaster walls, bricks, or stone. In assessing for mold there are many elements that we need to understand: One, is there mold and where is it? Two, how much mycotoxins are in the environment? Three, what conditions led to this situation?
What are the options for testing for mold?
When performing home mold testing for mold, there are four common options for testing mold: Air samples, ERMI, HERTSMI-2, and EMMA-mold count. I will detail out the strengths and weaknesses of all four of these options. The first test (air samples) is a direct assessment by counting spores on a microscope, and the following three of these tests are PCR (polymerase chain reaction) tests which look for DNA from fungal species in dust samples.
Air samples are a cheap way to test for mold spores. Using a vacuum-like device the spores are impacted unto a slide which has an adhesive attached. A lab then counts the number of spores on the slide and then multiples that number in relation to how much air was pulled through the device.
The ERMI test is based on data from the EPA and the 2006 HUD American Healthy home survey. There are 36 different molds in these tests. There are 26 water damage molds and 10 common molds. The benefit of this test is that the client receives a lot of data. The downside is that some remediators scare clients with results that show high amounts of common molds. The other downside is that in houses with both elevated, dangerous, and common mold it can artificially lower the causing score to be a normal range result.
HERTSMI-2 (Health Effects Roster of Type-Specific Formers of Mycotoxins and Inflammagens-2nd version) was developed by Dr. Ritchie Shoemaker to look at what he decided were the five most dangerous molds. The data from this report can be used to produce a score by using the spore counts from the five different species, i.e.: A spore count of Aspergillus versicolor of over 500 will be 10 points. Some individuals use a score of 5 and below as safe, while others use a score of 10. The big downside of the HERTSMI-2 is that many pathogenic species of mold are left out of this analysis, such as Penicillium. Ochratoxin A, the most common mycotoxin found in humans, is produced by species of Penicillium.
The fourth mold test is the EMMA-part 1. I call this part 1 because the EMMA is actually two tests, a mold test and a mycotoxin test (which will be discussed later in this blog). The EMMA is run by our company, RealTime Labs. The mold portion is currently 11 mold species; however, we are looking to add more.
Testing for mycotoxins is more important than just testing for mold
While home mold testing is essential, we believe that testing for mycotoxins is even more crucial and informative. Testing for mold is important and probably the most common way to test for home toxin issues. However, I would argue that testing for mycotoxins is more critical and more informative. Mycotoxins are the toxins produced by mold. When molds feel threatened, they produce these metabolites. The types of toxins produced are dependent on the species of mold, the environment the mold is growing, and the food source. As mentioned earlier, the EMMA [hyperlink to product page] is a combo test with two parts, and the second part is measuring mycotoxins. Mycotoxins, which are molecules, are over 1000 times smaller than spores. Mycotoxins have the capacity to infiltrate areas of a building that spores may be unable to reach. In many instances, especially with Stachybotrys, you will not find mold spores in the main living area, but you will find mycotoxins. This is because spores are multiple folds larger than mycotoxins and, in many instances, they are sticky. Spores can easily be trapped behind a wall but can still affect the inhabitants because of the toxins they release. In my experience, about 30% of the time a house will come back normal in mold spore counts but elevated with mycotoxins because of the size difference.
Besides testing your house, you should be looking into preventive measures. Measuring humidity and moisture are important elements in mold growth. This explains why we have the most mold clients in Florida, the Pacific Northwest (Seattle, Portland, Vancouver), and the Gulf of Mexico region (Houston and New Orleans), which are all high-humidity areas. Keeping your home between 25-55% humidity will maximize your health and minimize the threat of mold growth. However, I have consulted on mold cases from every state in the US because construction and maintenance issues are not tied to a particular geographic region. Studies indicate that moisture damage and mold in the main living areas of the house are the most correlated with adverse respiratory effects (Karvonen et al., 2015; Kirjavainen et al., 2016; Mustonen et al., 2016). In addition, moisture damage in the building is correlated with harmful outcomes for those that live/work in the building (Park, Schleiff, Attfield, Cox-Ganser, & Kreiss, 2004). One important number is the water activity (aw) ( aw x 100 = % relative humidity at equilibrium), and the longer a material’s aw is over 0.75, the greater the risk for fungal growth (Nielsen, Holm, Uttrup, & Nielsen, 2004).
If you have any questions, feel free to contact our team. We can provide insight into both clinical and home testing.
Note: The views in this blog are those of Dr. Pratt-Hyatt and not necessarily the views of RealTime Labs.
Karvonen, A. M., Hyvarinen, A., Korppi, M., Haverinen-Shaughnessy, U., Renz, H., Pfefferle, P. I., . . . Pekkanen, J. (2015). Moisture damage and asthma: a birth cohort study. Pediatrics, 135(3), e598-606. doi:10.1542/peds.2014-1239
Kirjavainen, P. V., Taubel, M., Karvonen, A. M., Sulyok, M., Tiittanen, P., Krska, R., . . . Pekkanen, J. (2016). Microbial secondary metabolites in homes in association with moisture damage and asthma. Indoor Air, 26(3), 448-456. doi:10.1111/ina.12213
Mustonen, K., Karvonen, A. M., Kirjavainen, P., Roponen, M., Schaub, B., Hyvarinen, A., . . . Pekkanen, J. (2016). Moisture damage in home associates with systemic inflammation in children. Indoor Air, 26(3), 439-447. doi:10.1111/ina.12216
Nielsen, K. F., Holm, G., Uttrup, L. P., & Nielsen, P. A. (2004). Mould growth on building materials under low water activities. Influence of humidity and temperature on fungal growth and secondary metabolism. International Biodeterioration & Biodegradation, 54(4), 325-336. doi:https://doi.org/10.1016/j.ibiod.2004.05.002
Park, J. H., Schleiff, P. L., Attfield, M. D., Cox-Ganser, J. M., & Kreiss, K. (2004). Building-related respiratory symptoms can be predicted with semi-quantitative indices of exposure to dampness and mold. Indoor Air, 14(6), 425-433. doi:10.1111/j.1600-0668.2004.00291.x
